
Department of Neurobiology
Research Building 17, Room 5.156
Route: 0642 | Email: kmkoss@utmb.edu
Research Interests
The Koss lab is based at the University of Texas Medical Branch (UTMB) in the Neurobiology Department and the Sealy Institute for Drug Discovery (SIDD), where our focus is on understanding and mitigating the inflammatory rejection of biomaterials through peptide bioengineering and discovery.
Dr. Koss’ research journey began with a foundation in chemical engineering, followed a doctoral and postdoctoral research that delved into neuroinflammation and biocompatibility within the nervous system. This work revealed a critical observation: the success or failure of implanted biomaterials hinges on their interaction with glial cells – primarily microglia (key players in tissue death and immunity) and astrocytes (central to the formation of problematic CNS scars).
While recognized for their role in rejection, these glial cells also possess crucial regenerative capabilities. My research aims to harness the beneficial potential of glia by:
- Developing strategies to target inflammatory responses to enhance cancer therapies and improve the acceptance of neural implants.
- Promoting regenerative processes in neuroinflammatory-related injuries.
To investigate glial reactivity, the Koss lab cultures microglia and astrocytes and uses them in a variety of tissue engineering applications including peptide discovery, neuroinflammatory phenotype development (healthy, acute, and chronic), 3D cell models for implant rejection, and paradigms for transplantation (limb transplantation).
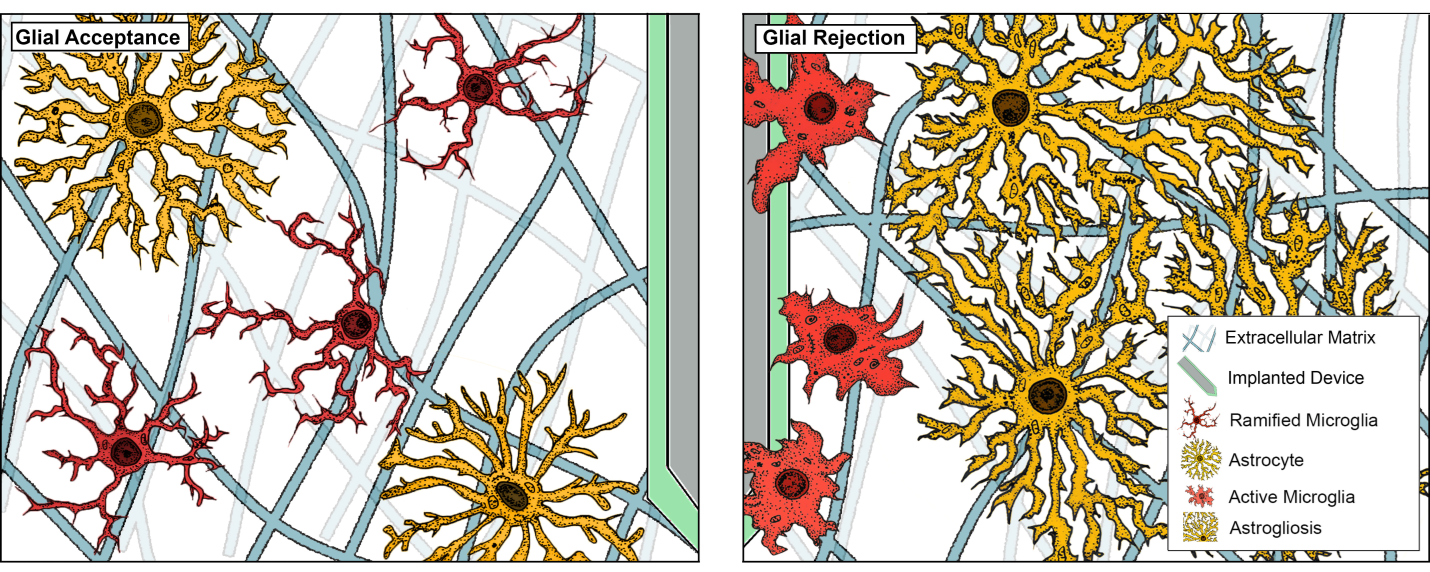
Rejection of implantable device/material in CNS. Shown here is an electrode being targeted by microglia, the resident immune cell, which subsequently recruit astrocytes to generate the glial scar (PMID: 30414483).
In most recent funding, we expanded our research to encompass vascular inflammation and allograft transplant rejection in various organs. Funded by the American Heart Association (AHA), we developed heparin-peptide conjugates to minimize post-surgical inflammation and promote wound healing in tissues associated with kidney, liver, and limb transplants. Funded by the Canadian Institutes of Health Research (CIHR), we developed wireless sensors to monitor transplant rejection in a hind limb model and are currently adapting this to monitor neuroinflammation and gliosis. Other areas of interest:
- Microglial mitophagy and peptide-mediated mitochondrial DNA delivery.
- Wireless electron-transfer in neural-interfacial peptide networks.
- Wireless biosensors to detect neuroinflammation and glial rejection.
- Peptide biomarker discovery in opioid-addicted microglia.
- Hyaluronic acid binding peptides and hydrogles for wound healing and implant acceptance
Recent Citations:
- Ameer, GA, Wertheim, JA, Jiang, B, K. Koss, KM, Heparin conjugated to collagen-binding peptides for targeting to biological and synthetic tissues, US12036234B2, 2024.
- White, MJV, Ozkan, M, Gomez-Medellin, JE, Rączy, MM, Koss, KM, Solanki, A, Zhang, Alpar, AT, Naved, BA, Wertheim, JA, Hubbell, JA, Blocking antibodies against integrin-α3, -αM, and -αMβ2 de-differentiate myofibroblasts, and improve lung fibrosis and kidney fibrosis. Scientific Reports. 2024. 14: 21623. DOI: 10.1038/s41598-024-70737-4. (PMID: 39284829)
- Hekman KE, Koss KM, Ivancic DZ, He C, Wertheim JA. Autophagy enhances longevity of induced pluripotent stem cell-derived endothelium via mTOR-independent ULK1 kinase. Stem Cells Translational Medicine 2022 Nov 18; 11:1151-1164. (PMCID:PMC9672854)
- Navel BA, Han S, Koss KM, Kondo MJ, Jiao-Jing W, Weiss C, Passman MG, Wertheim JA, Luo Y, Zhang ZJ. Multivariate description of gait changes in a mouse model of peripheral nerve injury and trauma. PLoS One 2025 Jan 7; 20(1):e0312415. (PMCID: PMC11706367)